Check out our new Augmented Reality Apps! Check out our Giants in Math and Science Series: Ada Lovelace • The First Coder |
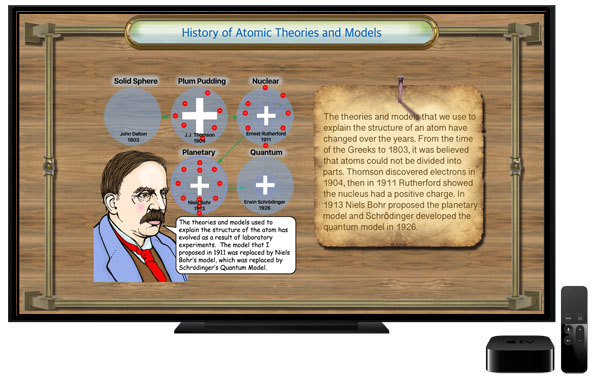
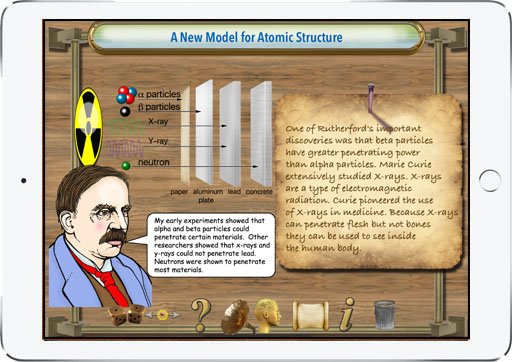
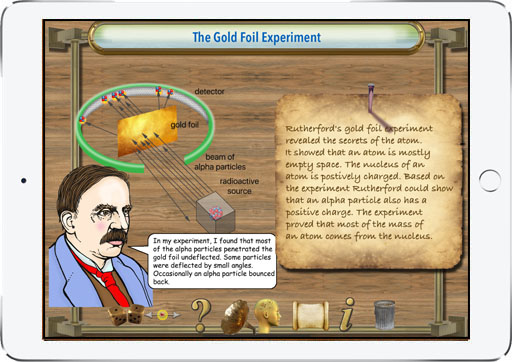
With this app students learn about the life and contributions to nuclear physics made by Ernest Rutherford. It discusses the early conceptual models for the structure of the atom and how Rutherford's work was a step toward the quantum model used by physicists today. Diagrams are used to illustrate key concepts. Rutherford proposed a theory to explain radioactivity. Radioactivity is the process by which certain elements, for example, uranium or radium, are in a constant state of decay. Rutherford identified the particles emanating from radioactive materials. He named them alpha and beta. The alpha particles are basically a helium atom. Beta particles are electrons. For his discovery Ernest Rutherford was awarded the Nobel prize in chemistry in 1908. The app presents a survey of the significant contributions that Ernest Rutherford made to further the development of modern physics. He was one of the first to reveal the structure of the atom and the nature of alpha and beta particles. Students will enjoy using this graphically illustrated, interactive learning tool. The timeline feature presents key events in the history of chemistry and physics and tapping on timeline points brings up descriptions of each of the milestones that led to our modern understanding of electromagnetism. A quiz function helps students demonstrate their comprehension of the reading material. Key concepts related to nuclear physics are shown in colorful illustrations. We hope this app will inspire students to study chemistry, physics and science in general. The app shows that Ernest Rutherford certainly deserves a significant place in the history of nuclear physics.
|
Special features make using Ernest Rutherford • Nuclear Physicist fun: 1. Tap buttons to learn about the major events in his life. 2. interesting facts are presented using colorful charts. 3. Tap the quiz or game buttons to practice what you've learned. 4. Read about some of the significant events in the history of chemistry and physics and the important contributions that Ernest Rutherford made during his lifetime. 5. Sound effects make the interaction even more fun. 6. Available for iPad and Apple TV devices. |
 |
Available now for Apple TV Bring the excitement of exploring concepts related to nuclear physics and contributions of Ernest Rutherford to your classroom or living room with the Apple TV version of the Ernest Rutherford - Nuclear Physicist tvOS app.
|
 |
$1.99 |